Estimated time: 20 minutes. No prior knowledge needed.
Watch the video, then follow the instructions.
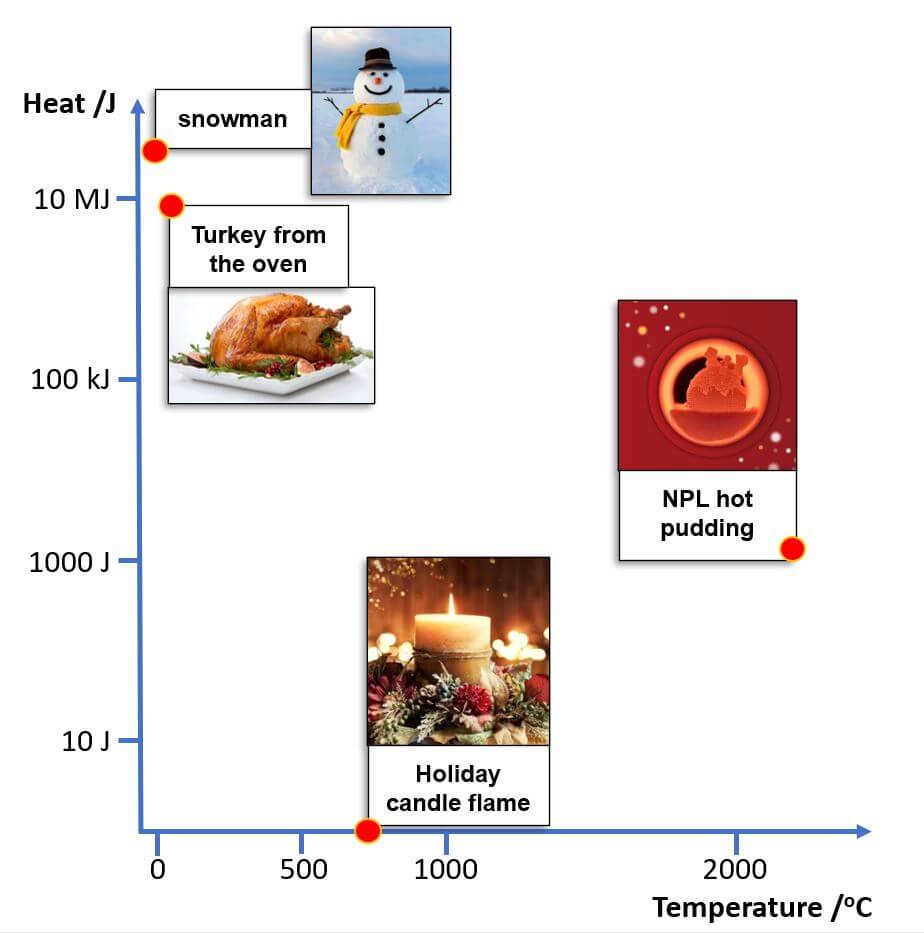
Choose an object. At the time of writing it’s Christmas so ours are seasonal. It’s best if your object is made of just one material (like iron or water), though you can simplify (e.g. a person = 100% water).
Measure or estimate the mass (in kg) and temperature of your object. Convert °C temperature value to K (kelvin) by adding 273.
Find your material’s ‘specific heat capacity’ value. As living things are mostly water, for them, you could use the value for water = 4,200 J/kg/K. (engineeringtoolbox.com) (material-properties.org)
Calculate the heat (total thermal energy) of your object
= mass (in kilogram) x temperature (in kelvin) x specific heat capacity.
There are examples in the downloadable worksheet.
Repeat for other objects and share your results with us, or compare them with our festive object heat vs temperature plot below.
Measurement science, maths, specific heat capacity.
Temperature measures hotness/coldness of a body while heat is a type of energy.
We calculate heat (thermal energy, unit joule (J)) transferred to change an object’s temperature. Temperature change depends on the object’s mass and a property called ‘specific heat capacity’.