Non-viral encapsulation systems, such as liposomes, are sought after for a number of biological applications, ranging from drug and gene delivery to vaccines. The characterisation of their quality attributes is critical in ensuring reproducible manufacturing, supporting regulatory approval and improving clinical performance. The visualisation and characterisation of these delivery systems, which are built, perform and function at the nanoscale, is challenging due to their size and soft nature. There is therefore the need to develop high resolution imaging measurements, capable of probing soft encapsulation systems in their native state and with nanoscale precision to:
Cryo-transmission electron microscopy (cryo-EM), with sub-nanometer resolution, is the technique of choice to demonstrate structural parameters of soft and biological materials, including size, ultrastructure and morphology. In a recently completed A4I project, NPL applied its expertise in cryo-EM to assess the liposomal formulations of crystalline anticancer drugs, namely Doxil® and Onyvide®. NPL used cryo-EM imaging, complemented by a suite of analytical techniques for nanoparticle analysis in solution, to measure the key quality attributes of these formulations. Cryo-EM imaging provided invaluable information, not only on size distribution, but also on morphology of the formulated drugs and the loading efficiency of crystalline materials, in particular in relation to the ratio of loaded and unloaded liposomes (i.e. full/empty ratios). This information was invaluable to support Oxford HighQ, a UK-based company developing novel instrumentation for particle analysis. Their innovative technology is based on the use of optical microcavities to measure, amongst other parameters, drug loading and size distributions. Cryo-EM provided quantitative information on the thickness of the liposome lipid bilayer (3-4 nm) which informed the optimisation of the modelling employed by Oxford HighQ for particle analysis. This work was crucial to demonstrate the applicability of optical microcavities to the analysis of liposomal drug delivery systems.
Emiliana De Santis, Senior Research Scientist, who led the cryo-EM activities said: “It was an exciting collaboration. Not only were we able to visualise the liposomes and their encapsulated drug, but the cryo-EM imaging also highlighted structural and morphological changes associated to drug-loading with nanoscale precision.”
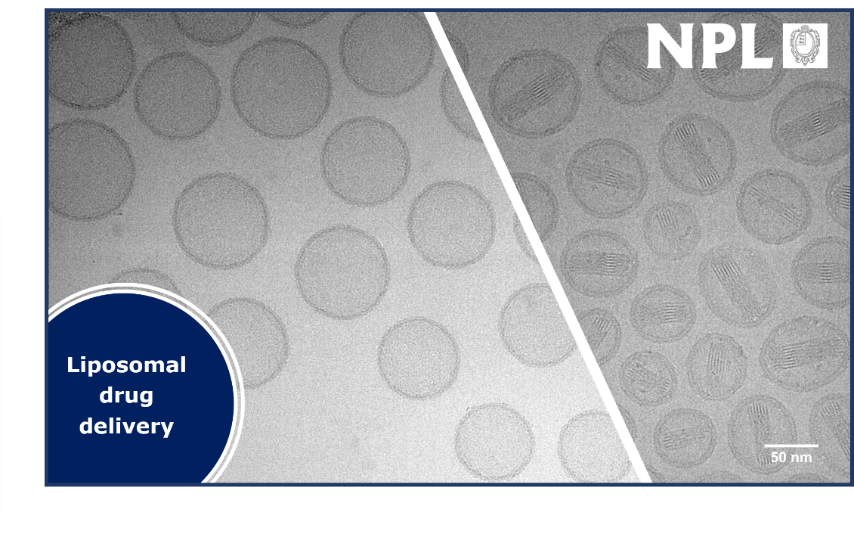
The work helped Oxford HighQ to optimise the modelling of their particle analysis instrumentation and allowed the company to expand the application range of their novel technology.
We demonstrated the impact of high resolution cryo-EM and validated that it is a useful tool for providing invaluable structural information on liposomes, such as size and morphology, concomitantly. The work also highlighted the key role cryo-EM plays in supporting, benchmarking and validating novel analytical technologies for the analysis of soft nanoparticles, such as viral and non-viral vectors. This will help progress applications such as gene delivery and vaccine development, including in commercial COVID-19 vaccines.
The ability to visualise encapsulating systems is essential for ensuring reproducible manufacturing, supporting regulatory approval and improving clinical performance in these applications.
NPL’s team provided us with new and important insights that are key for the development and validation of our novel technology. The collaboration generated a useful dataset to inform the optimisation of our technology and expand its application range.
Aurélien Trichet - CTO of Oxford HighQ