Immunoassays (IAs) are highly sensitive techniques which are widely used to detect or quantify a specific molecule in a body fluid sample. The technique utilises highly specific binding of the target molecule to a conjugated antibody to signal its presence. During the conjugation stage of assay development, the antibody may be damaged and that, in turn, impacts assay specificity and performance.
Developments in IAs and advances in complementary technologies have paved the way to several point-of-care devices. They can be used to achieve the low detection limits which are vital for early detection of cancer, post-treatment relapse or pregnancy tests. There is considerable research underway to exploit these IA technologies to enable personalised, mobile healthcare and empower patients to monitor their own health.
Fleet Bioprocessing Ltd are at the forefront of protein conjugation technologies and offer services to help assay development companies build increased specificity and performance into their antibody binding assays. Scientists at Fleet Bioprocessing worked with NPL to deliver faster and cheaper, non-destructive analyses to understand assay performance early in the production cycle. Characterising performance earlier in the assay production process drives significant cost savings and delivers a higher quality product.
During the conjugation reaction a molecule, such as biotin, is attached to an antibody and, while the overall structure of the conjugated antibody is not affected by the conjugation process, subtle changes in structure may impact antibody reactivity or stability. Fleet Bioprocessing generated panels of antibody conjugates and established assay formats to allow the behaviour of these conjugates to be quantified. However, it was found that a conjugate performing well in one assay format did not necessarily work well in another, demonstrating clear differences in behaviour within each panel when tested with the relevant assays.
NPL used Circular Dichroism (CD) spectroscopy to predict immunoassay performance by determining structural changes due to the conjugation process. Though ambient CD is powerful in capturing structural changes, it was not effective in differentiating subtle structural changes between conjugates which depend on the type and number of labels incorporated and conjugation chemistry. Therefore, NPL evaluated the stability as the temperature was gradually increased and determined the temperature at which a structural change was observed.
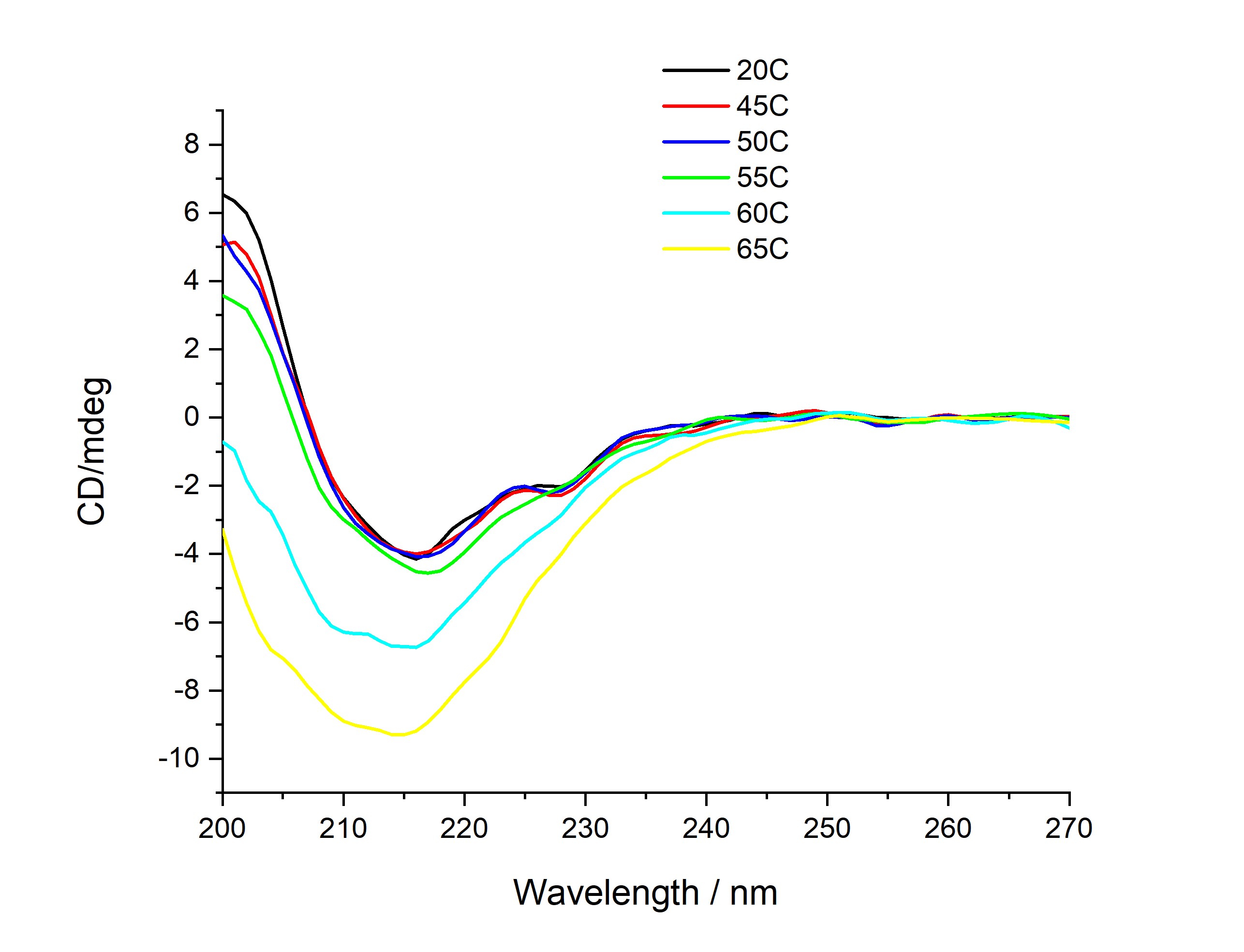
CD-thermal melt of antibody ligand conjugate
Differences in spectra were observed at elevated temperatures due to loss of structural integrity of a given antibody conjugate. The onset temperature at which the changes started to occur is deemed to be an important property in predicting the performance of the antibody conjugates. Low onset temperatures signal unstable conjugates and those conjugates which required high temperatures to loose structure are deemed stable.
This demonstrated that thermal denaturation of antibodies monitored by CD spectroscopy provides a cheaper and faster method for predicting IA performance than alternative techniques such as mass spectroscopy or NMR (nuclear magnetic resonance) which can be expensive and time consuming.
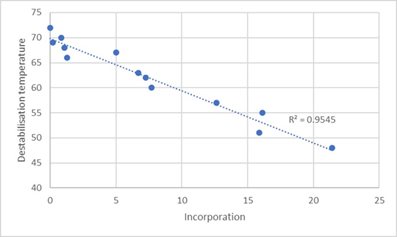
Correlation with destabilisation temperature versus ligand incorporation level
The application of CD as a rapid quality probe for the performance of immunoassay conjugates enabled Fleet Bioprocessing to confirm the suitability of a conjugate, without the need for expensive and time consuming, destructive testing. This streamlined the assay development process by about 20%, which in turn improved productivity and delivered a significant competitive advantage through enhanced characterisation data.
"CD is generally used for rapid comparison and characterisation of protein structures," explains Jascindra Ravi, Higher Research Scientist. "However in this study a new application was proposed. It was a challenge to come up with a fast and cost-effective bioanalytical method to predict conjugated antibody performances."
The work and understanding of assay performance delivered through the project will enable other organisations to utilise the CD spectroscopy analyses to understand assay performance and can be extended to other compounds.
This study started with a three month Analysis for Innovators (A4i) funding programme, and the results of the project resulted in a follow-on study for another year.
We were very lucky to be able to access BioMet’s bioanalytical expertise on this project. Once they had understood the problem we were trying to address they were able to suggest a number of analytical techniques that we were not familiar with, and one of these – thermal CD spectroscopy – proved to correlate well with antibody damage during conjugation.
Dr Alaistair Dent, Managing Director and Quality Director - Fleet Bioprocessing