Oncoinvent is a small life sciences company, delivering innovative cancer treatment by injecting radiopharmaceuticals directly into affected areas. This technology has the potential to allow more targeted treatment to regions of organs and the body which are difficult to access, while minimising harmful radiation doses. Oncoinvent’s products deliver treatment using radiopharmaceuticals incorporating the alpha-particle emitting radium-224 (Ra-224). The challenge with this approach is to quantify the amount of radioactivity administered to the patient and ultimately understand the dose delivered to the tumour.
Oncoinvent are developing Radspherin®, an inorganic microsphere containing Ra-224, which has been shown to cause a reduction in tumour cell growth in model systems and significantly increase survival rates. It has the potential to treat several forms of metastatic cancer.
The complexity of the decay chain and ingrowth equilibrium of Ra-224, and the decayed radioactive species, means significant expertise is required to develop primary and secondary standards and enable in-situ measurement traceability. It is vital that accurate radioanalytical procedures are in place for the quality assurance checks of the product which will enable preclinical and clinical trials.
In addition, the microparticle delivery system for Ra-224 requires morphological and chemical characterisation to assess their stability during sterilisation, shipping, batch consistency and absence of contaminants.
With its expertise in nuclear metrology, NPL has supported Oncoinvent by performing the world first primary standardisation of Radium-224 decay, enabling further studies of relevant nuclear decay data. The radiochemistry team at NPL also developed a highly efficient procedure for impurity checks which allows the detection of trace levels of impurities of other alpha-particle radioemitting nuclides.
NPL used its particle technology expertise to measure the morphology, crystal phase and surface chemistry of different batches of the inorganic delivery particles. The resulting information enabled Oncoinvent to select the materials and processes that ensure product uniformity and reproducibility.
Quality and reproducibility of data is absolutely key for medical treatments, and our multi-disciplinary approach enabled Oncoinvent to have confidence in the nuclear data and the particle characterisation.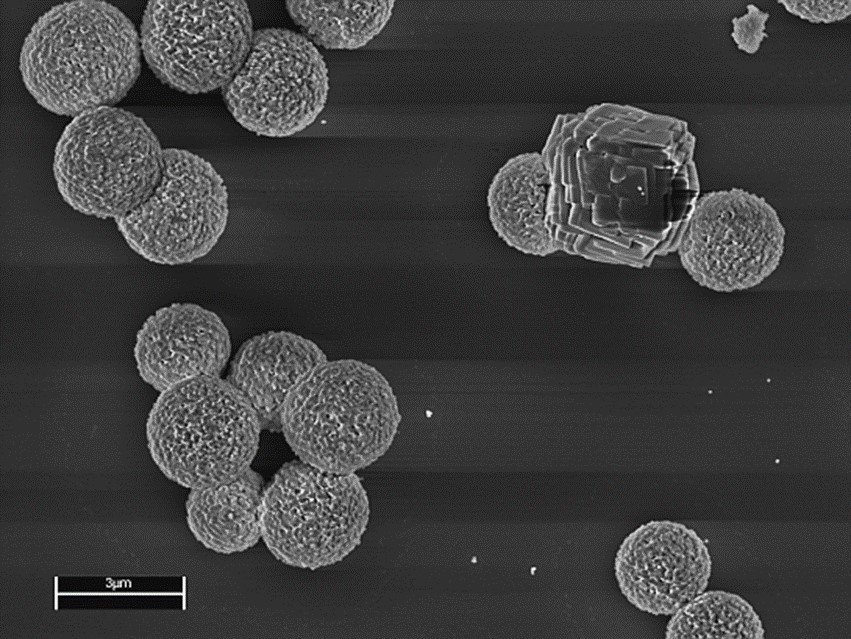.jpg?width=350&height=261)
NPL developed primary standards for Ra-224, which had not previously been established for this complex element. This work underpins future secondary standardisations of this radionuclide in a cost effective and traceable manner. This will be used by Oncoinvent as well as being more widely available to the nuclear medicine community.
The collaboration with NPL enabled Oncoinvent to apply for regulatory approval to move this emerging radiopharmaceutical treatment to pre-clinical trials. The first clinical trials for Radspherin® will be for the treatment of peritoneal carcinomatosis originating from ovarian cancer, one of the most serious complications of gastrointestinal and gynaecological malignancies.
Traceability to a Metrology Institute like NPL, is of great importance for Oncoinvent. Accurate measurement of radium-224 in production, quality control of the dose administered to the patient is vital. This ensures the safety and effectiveness of treatment using Radspherin.
Gro Elisabeth Hjellum - Quality Control team at Oncoinvent